INTRODUCTION
Most pharmaceutical growth stories are told at launch. The molecule is new. The data is fresh. The curve is steep. What gets less attention is what happens ten years later, when the science still holds, the prescriptions still come in, but the competitive edge feels thinner.
That is where lifecycle strategy becomes a profit discipline. And increasingly, one of the most effective lifecycle moves is also one of the most practical: delivery-format reformulation.
Orally disintegrating and lyophilized tablets (ODTs) are increasingly part of that conversation because they give established brands a way to evolve without starting over.
The interest is visible at the market level.
Industry research firms project the global ODT market to roughly double over the coming decade, driven largely by reformulations and line extensions rather than brand-new molecules. That growth pattern tells its own story: lifecycle teams are actively choosing delivery upgrades as a value strategy.
The Second Curve Is Built on Practical Moves
Mature brands rarely get a second growth curve from dramatic reinvention. They get it from smart, practical moves that improve fit, use, and positioning.
"Innovator and generic companies are looking for incremental innovation for products already in the market, differentiation that is commercially realistic," says Kamal Preet, Vice President, InstaPill.
Reformulation sits squarely in that category. An ODT version of an existing product gives a portfolio new commercial angles:
- ● A differentiated format
- ● A refreshed brand story
- ● A visible product upgrade
- ● A reason to re-engage channels and prescribers
- ● A new tier in the same franchise
All of this happens while the core molecule and the trust built around it stay intact. Teams work with known products and known safety, and development programs stay focused.
Delivery Format as Brand Refresh
Lifecycle strategy often looks for ways to make a mature brand feel current again. A packaging redesign can help with that. New messaging can help. Delivery format goes further by changing the product experience itself.
An ODT format introduces a clear, user-visible difference:

Those features translate directly into marketing language and channel storytelling. They support premium variants, segment extensions, and portfolio refresh campaigns without rewriting the science. The commercial benefit is easy to explain because the upgrade is tangible.
Field teams can show it. Patients can feel it. Buyers can recognize it.
Why 505(b)(2) Pathways Strengthen the Business Case
Another reason ODT reformulation appears frequently in lifecycle pipelines is regulatory economics. In the U.S., many reformulated products can follow the 505(b)(2) pathway, which allows sponsors to rely partly on existing safety and efficacy data. Comparable hybrid pathways exist in other major markets.
In portfolio strategy, this is often described as low-risk innovation: development built on known molecules, supported by prior evidence, and structured through defined regulatory pathways. That combination aligns well with lifecycle capital filters and timeline expectations.
Where Adherence Enters the Lifecycle Picture
Adherence rarely sits at the top of a lifecycle slide, yet it shows up at the bottom of every revenue curve. Persistence, refills, and continuation shape the long tail of brand performance.
Delivery format influences daily dosing behavior in practical ways. Easier administration reduces friction around taking the medicine. Over time, that supports steadier use patterns.
Industry platform leaders often link ODT reformulation to improved compliance, particularly in patient groups where swallowing difficulties affect dosing adherence. In lifecycle terms, adherence works as a quiet multiplier. Small improvements in consistency accumulate across large patient populations.
Route-Based Differentiation Is Showing Results Across Portfolios
Route-of-administration upgrades are producing lifecycle wins across multiple portfolio types, from consumer-facing OTC brands to long-duration chronic therapies and specialist segments. The commercial logic is consistent: delivery experience becomes part of brand value
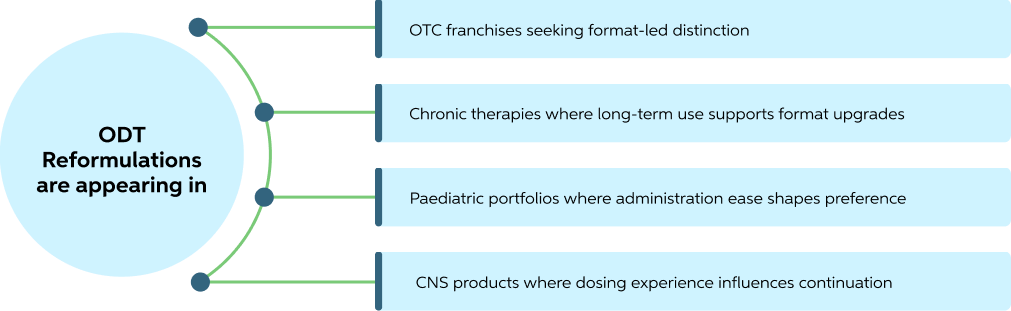
These are lifecycle-driven moves, designed to extend relevance, refresh positioning, and create a product-level talking point without altering core pharmacology.
In Summary
Blockbuster launches create momentum. Lifecycle strategy creates longevity.
This advantage increasingly comes from design choices that compound over time. Reformulation through ODT formats represents a disciplined form of incremental innovation: product-visible, commercially meaningful, and operationally grounded.
In mature portfolios, growth increasingly follows intelligent evolution. Delivery format now holds a defined place in that growth equation.
Key Takeaways
● Lifecycle growth increasingly comes from reformulation, not reinvention. Mature brands are extending commercial relevance through delivery-format upgrades that create product-visible differentiation without resetting scientific foundations.
● Extended-release formats, fixed-dose combinations, and flavor variants remain useful, yet route of administration innovation is emerging as a higher-impact differentiator in crowded categories.
● Known molecules, defined regulatory pathways, and contained development scope make ODT pathways attractive as structured incremental innovation programs.
● Reformulated delivery formats often progress through established regulatory frameworks that leverage existing safety and efficacy data, strengthening the business case.
● Delivery format influences persistence, and persistence supports lifecycle value. Ease of administration contributes to steadier real-world use, making format innovation a practical lifecycle multiplier over long treatment horizons.
References:
- Ken Research. (n.d.). Global Orally Disintegrating Tablets Market – By Type, By Age Group, By Distribution Channel, By Geography, Competition Forecast & Opportunities, 2029. Retrieved October 14, 2025
- Persistence Market Research. (n.d.). Pharmaceutical Excipients Market.
- Drug-Development & Delivery. (n.d.). Formulation Forum: Orally Disintegrating Tablets.

